The organic chemicals present in indoor environments come from a variety of sources, including outdoor air, emissions from indoor materials, building occupants and their activities, (such as cooking, cleaning, and smoking,) and chemical reactions that occur in air and on surfaces including the skin and clothing of occupants. These chemicals can significantly affect the quality of indoor air.
In this project, we conduct pilot studies in a variety of locations to investigate the sources and processes that influence the composition of organic chemicals in indoor environments. Study sites include classroom, dining, and athletic facilities, an art museum, and offices and laboratories at the University of Colorado-Boulder. We aim to address the question: What are the sources, sinks, and chemical transformations that determine the composition of organic chemicals in indoor environments, and can we describe these processes at the molecular level? In the course of answering this question, we hope to develop a deeper understanding of how different indoor environments function. Although there have been many studies of the factors that influence the chemical composition of indoor air, few have attempted to understand the relevant chemistry at the molecular level. Most have relied instead on a large body of knowledge available on the chemistry of outdoor air or other systems, which is often insufficient or not applicable to indoor environments. It is due to this absence of fundamental knowledge specific to indoor air chemistry that the Sloan Foundation’s Chemistry of Indoor Environments (CIE) program includes among its basic scientific research and exploration goals a desire to understand and predict how materials, occupants, and physical and chemical processes that occur in built environments influence the chemical composition of indoor air.
We combine laboratory and field experiments to answer five main research questions:
- What are the similarities and differences in the organic chemical composition of gases, particles, and surfaces in some commonly encountered indoor environments?
- What are the relative contributions of outdoor air, surface processes, human occupants, and other sources to the organic chemical composition of gases and particles in the studied indoor environments?
- What are the effects of organic gases, oxidants, acids, humidity, light, and temperature on gas, particle, and surface composition indoors?
- What are the potential effects of organic compounds emitted by human occupants–either directly or as a result of reactions on skin or clothing–on the composition of indoor air?
- Can mechanisms be developed to explain chemical compositions and processes occurring indoors at the molecular level?

The University of Colorado Art Museum
In the summer of 2017, we conducted a field campaign (ARTISTIC) at the University of Colorado Art Museum, a relatively clean and controlled environment. Future field campaigns will take place in the University of Colorado Athletic Center, and the University of Colorado Dining Center.
Measurements and Instrumentation:
We employ a suite of state-of-the-art instruments to analyze various components of indoor environments. To measure organic gases, we use a Proton Transfer Reaction Mass Spectrometer (PTR-MS) and a FIGAERO-CIMS, which allow for the detection of organic gases over a range of volatility. To measure aerosol particles, we use both the FIGAERO-CIMS, which collects particles on a filter prior to analysis, and the High Resolution Aerosol Mass Spectrometer (HR-AMS). We also monitor aerosol size distributions using a Scanning Mobility Particle Sizer (SMPS), and nitrogen oxides and ozone are measured using standard NO-NO2-NOx and O3 analyzers.
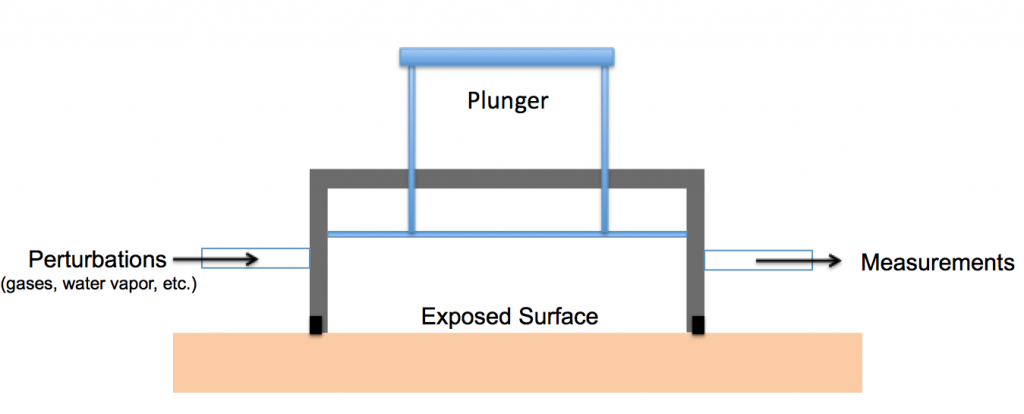
We have also built and characterized a wall reactor to explore chemistry occurring on indoor surfaces. Briefly, the wall reactor cell is sealed to a surface, such as a painted wall or tile floor, and the air above that surface can be perturbed by adding different classes of organic gases, atmospheric oxidants, water vapor, and acids to the reactor. The surface can also be exposed to UV or visible light. Changes in the chemical composition of gases and particles sampled from the wall reactor and the exposed surfaces can then be determined in real time and from offline analysis of collected samples.
Principal Investigators:
- Dr. Paul J. Ziemann (PI): Professor, Department of Chemistry & Biochemistry, CIRES Fellow; University of Colorado, Boulder
- Dr. Jose-Luis Jimenez (co-PI): Professor, Department of Chemistry & Biochemistry, CIRES Fellow; University of Colorado, Boulder
- Dr. Shelly Miller (co-PI): Professor, Department of Mechanical Engineering; University of Colorado, Boulder